Solensia is a once-monthly, injectable monoclonal antibody therapy for the control of feline osteoarthritis (OA) pain
- Controls the pain of OA in cats, helping to improve their mobility, comfort and well-being1
- 77% of cat owners reported improvement in signs of pain when their cats were treated monthly with Solensia in a 3-month study†
- Solensia was found to be well tolerated in field studies that included cats identified as IRIS stage 1 or 21†‡


†67% of cat owners experienced improvement in signs of their cat's pain in the placebo group.
‡Total N = 131 of 267 treated cats were identified as IRIS stage 1 or 2
Read more about feline OA pain signs and treatment
Read more about feline OA pain signs and treatment



Get updates about Solensia
Be the first to know about product news, useful resources and more.




One monthly injection controlled pain, increasing mobility1

Solensia is administered monthly at the veterinary practice
ABOUT SOLENSIA
A Breakthrough in OA Pain Control



Solensia is the first and only once-monthly monoclonal antibody therapy that controls feline osteoarthritis (OA) pain. Solensia is administered in the clinic.
Targets nerve growth factor (NGF) to control pain
77% of cat owners reported improvement in signs of pain when their cats were treated monthly with Solensia in a 3-month study†
Controls the pain of OA in cats, helping to improve their mobility, comfort and well-being1
Functions like naturally occurring antibodies
†67% of cat owners experienced improvement in signs of their cat's pain in the placebo group.
Prepare your practice for Solensia
View shareable client resources
MECHANISM OF ACTION
Solensia Works Differently Than NSAIDs



Solensia is a monoclonal antibody (mAb)
- It is a highly specific protein targeting molecules involved in disease3
- It functions like naturally occurring antibodies—it is eliminated via normal protein degradation pathways, with minimal involvement of the liver or kidneys4
Solensia binds to Nerve Growth Factor (NGF), and its action helps to:5
- Control pain
- Limit the release of proinflammatory mediators and more NGF
- Lower neurogenic inflammation
In osteoarthritic joints, NGF is elevated.6 Solensia reduces the negative impact NGF has in osteoarthritic joints.
See how it works
EFFICACY
Efficacy Your Cat Patients Need
Reducing pain helps improve the quality of life for your cats.7 Solensia has been demonstrated to effectively control OA pain with a monthly injection.
In clinical field studies, Solensia effectively controlled pain associated with feline osteoarthritis, and is delivered in-clinic as a subcutaneous (SC) injection.
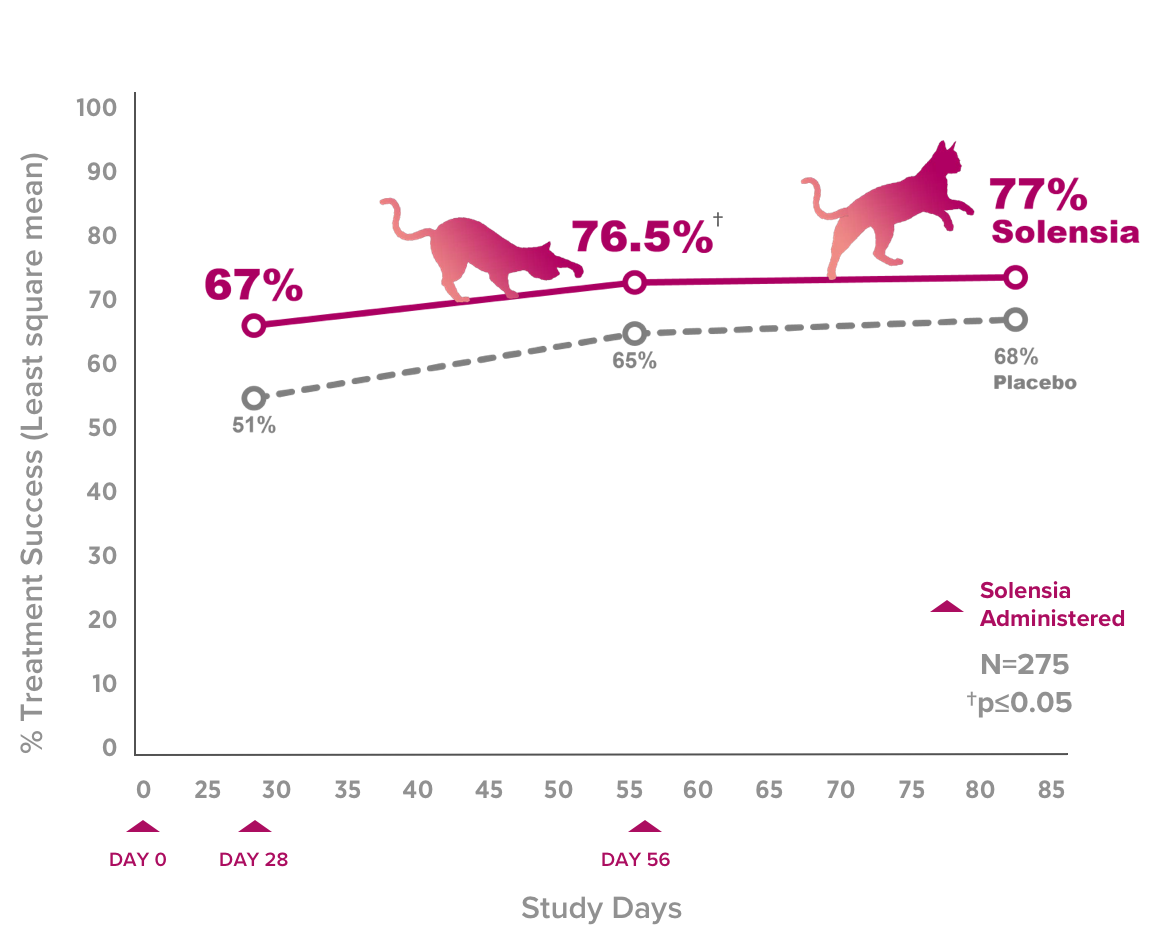


Successful Pain Control†
Three out of four cats benefitted from successful pain control over 3 months, in a pivotal trial using client reported improvement.**



Helps to Improve Mobility1
A pilot study found 12.9% greater activity in cats administered Solensia versus a placebo. This increase in activity was demonstrated in the first 3 weeks of the study in cats with DJD-associated pain using objective (and subjective) measurement.
**Client-specific outcome measures (CSOM) is a tool used to assess pain and pain treatment. It measures the impact of pain on the individual cat, including physical activity level, sociability, and quality of life.
†67% of cat owners experienced improvement in signs of their cat's pain in the placebo group.
SAFETY
Safety for Cats
Functions like naturally produced antibodies
Solensia is eliminated via normal protein degradation pathways—just like naturally produced antibodies—with minimal involvement of the liver or kidneys4
Solensia was shown to be well tolerated
The most commonly reported adverse events in a field study were vomiting and injection site pain
Well-tolerated at 5X the label dose
Clinical safety studies showed that Solensia given at 5X the highest label dose every 4 weeks for 6 months was shown to be well-tolerated
Allows more cats to be treated for longer
In clinical studies, Solensia was demonstrated to be a long-acting and well-tolerated pain medication, allowing more cats with OA pain to be treated for longer durations
DOSAGE & STORAGE
Take Control of Managing OA Pain
Dose:
1 mL for cats
5.5 - 15.4 lbs
2 mL for cats
15.5-30.8 lbs



Administration:
- Cats should be dosed by weight range according to the dosing chart
- Cats are given the full content of 1 or 2 vials based on body weight to target a minimum dosage of 0.45 mg/lb (1.0 mg/kg) body weight, administered subcutaneously once a month. Aseptically withdraw the total dose into a single syringe and administer immediately
- If a vaccine is to be administered at the same time as treatment with frunevetmab, the vaccine should be administered at a different site to that of frunevetmab administration
- Avoid excessive shaking or foaming of the solution
Storage:
- Store upright in a refrigerator, between 35° to 46°F (2° to 8°C). Do not freeze
- Protect from light
- Once opened, contents of the vial should be used immediately and any remaining solution should be disposed



Read Dosing and Administration Guides



“Once a month is better than trying to give my cat something once a day.”
“This is great, it’s once-a-month, less stress than two times a day.”
– Pet Owner Reactions to Product Profile2
Insert text here...

View tips for examining cats for OA

Find additional resources for OA pain

Get an OA checklist for your clients
References
- Gruen ME, Myers JAE and Lascelles BDX (2021) Efficacy and Safety of an Anti-nerve Growth Factor Antibody (Frunevetmab) for the Treatment of Degenerative Joint Disease-Associated Chronic Pain in Cats: A Multisite Pilot Field Study. Front Vet Sci. 8:610028. doi: 10.3389/fvets.2021.610028.
- Zoetis Market Research: KG MarketSense, 2018.
- Olivry T, Bainbridge G. Advances in veterinary medicine: therapeutic monoclonal antibodies for companion animals. Clinician’s Brief. Published March 2015.
- Keizer RJ, Huitema AD, Schellens JH, Beijnen JH. Clinical pharmacokinetics of therapeutic monoclonal antibodies. Clin Pharmacokinet. 2010;49(8):493-507.
- Enomoto M, Mantyh PW, Murrell J, Innes JF, Lascelles BDX. Anti-nerve growth factor monoclonal antibodies for the control of pain in dogs and cats. Vet Rec. 2019;184(1):23.
- Isola M, Ferrari V, Miolo A, Stabile F, Bernardini D, Carnier P, Busetto R. Nerve growth factor concentrations in the synovial fluid from healthy dogs and dogs with secondary osteoarthritis. Vet Comp Orthop Traumatol. 2011;24(4):279-284.
- Monteiro BP, Steagall PV. Chronic pain in cats: Recent advances in clinical assessment. Journal of Feline Medicine and Surgery. 2019;21(7):601-614. doi:10.1177/1098612X19856179.
SLN-00004R4
IMPORTANT SAFETY INFORMATION:
See full Prescribing Information. For use in cats only. Women who are pregnant, trying to conceive or breastfeeding should take extreme care to avoid self-injection. Hypersensitivity reactions, including anaphylaxis, could potentially occur with self-injection. Solensia should not be used in breeding cats or in pregnant or lactating queens. Solensia should not be administered to cats with known hypersensitivity to frunevetmab. The most common adverse events reported in a clinical study were vomiting and injection site pain.



